The Quest for Oral GLP-1s
In a recent survey, three-in-four respondents said they would prefer a once‑daily oral pill over a weekly injection of GLP-1s. So why aren't there more oral options?
By Dr. David S. Kim
Researchers have known that the pancreas secretes several peptide hormones critical to metabolic regulation, most notably insulin and glucagon, since the early 1900s. With the advent of recombinant peptide synthesis in the 1970s, scientists not only began working on isolating the gene responsible for insulin production but fabricating it outside the body. In 1978, scientists at Genentech turned bacteria into factories able to transform sugar into insulin by cutting the insulin gene from the human genome and pasting it into the genome of E. coli. This breakthrough allowed peptide drugs to be synthesized at mass scale, without needing to be harvested from animal pancreases, and gave birth to the biotechnology industry.
The role of glucagon and the gene encoding it, however, remained elusive. Then, in 1982, Joel Habener and colleagues at Massachusetts General Hospital (MGH) published a paper indicating not only that they had located the human glucagon gene, but that it actually encoded three separate peptides, two previously unknown. Those two unknowns were labelled as glucagon-like peptides: GLP-1 and GLP-2.
Svetlana Mojsov, a peptide chemist also working at MGH, managed to synthesize a truncated, but active, form of GLP-1. In 1987, her paper with Habener and Daniel Drucker showed that GLP-1 stimulates glucose-dependent insulin secretion in the pancreas, suppresses glucagon release, and slows gastric emptying, all of which stabilize blood sugar levels after eating.
While these effects initially made GLP-1 seem a promising candidate for drug development, the earliest clinical studies from 1992 and 1993 revealed that GLP-1’s half-life in the body was disappointingly short. A proteolytic enzyme, dipeptidyl peptidase-4 (DPP-4), binds to and destroys GLP-1 molecules in the blood in minutes.
Hope for boosting GLP-1’s half-life in the body came in the 1990s, when endocrinologist John Eng, at the Bronx Veteran Affairs Hospital, noticed something curious about the Gila monster while researching snake and lizard venom effects on the pancreas. Specifically, he was struck by how this desert lizard eats extremely infrequently, yet somehow maintains a stable blood sugar level for months.
Eng found that Gila monsters secreted a salivary venom peptide, now known as exendin-4. Structurally similar to GLP-1 but unaffected by DPP-4, this peptide lingers in the blood for hours rather than minutes. Exendin-4 thus became the molecular blueprint for exenatide, the first twice-daily injectable GLP-1 receptor agonist, approved by the FDA in 2005.

For many years after exenatide’s approval, GLP-1 receptor agonists were used primarily as diabetes medications. Practitioners and patients noticed, however, that taking such medicines produced weight loss as a side effect. During this time, researchers also found GLP-1 receptors in the brain and pancreas, particularly in areas controlling appetite and reward. And in 2021, the results from a series of five STEP trials (Semaglutide Treatment Effect in People with obesity) demonstrated marked weight loss even in individuals without type 2 diabetes (T2DM): Phase 3, for example, showed an average loss of about 15 percent over 68 weeks, more than double achieved by any prior obesity drug. In 2023, a trial of 17,604 adults (SELECT) with obesity and without T2DM, followed for over three years, showed a 20 percent reduction in major adverse cardiovascular events.
In response, GLP-1 prescriptions rose sharply. According to CDC data, the share of adults with type 2 diabetes using GLP-1 injectables more than doubled — rising 155 percent between 2018 and 2022 — while overall spending on these medications climbed more than 500 percent between 2018 and 2023.
The majority of patients continue to inject these drugs rather than take them orally. But a 2020 survey of 600 patients found that 76.5 percent would prefer a once‑daily oral dose over a once-weekly injection of GLP-1. Significant funding is going into R&D to launch an oral alternative that performs as well as an injectable version. While oral formulations are beginning to enter the market, so far they remain less effective and more expensive to manufacture. Why?
To the Stomach, All Peptides Are Food
Peptide drugs like GLP-1s are made up of a chain of amino acids chemically bonded together. For a GLP-1 molecule taken by mouth to reach its target, it must first pass through the stomach, where hydrochloric acid and peptidase enzymes cleave these amino acid bonds. Most ingested peptides are simply broken down into their constituent amino acids and absorbed as nutrition, indistinguishable from the protein in food.
The small percentage of GLP-1 molecules that might survive this journey and arrive into the intestines face a second gauntlet. Once in the blood capillaries, DPP-4 quickly degrades the molecule, and the drug is excreted by the kidneys.
Biochemical engineering, however, has allowed drugs like semaglutide to survive longer in the blood. First, an amino acid substitution stops DPP-4 from destroying GLP-1. This is achieved by swapping the alanine amino acid at position 8 in the peptide backbone for alpha-aminoisobutyric acid (Aib), a modified amino acid that DPP-4 cannot recognize or cleave. As a result, this modified form of GLP-1 is not degraded so swiftly.

Yet even a DPP-4-resistant peptide is small enough to be filtered by the glomeruli in the kidneys into the urine. Therefore, the chemists behind semaglutide made a second modification: they fused a fatty acid chain to the peptide in a process called lipidation. This causes the drug to bind to human serum albumin, a large transport protein abundant in the blood, with a natural half-life of approximately 20 days. The drug effectively co-opts albumin’s stability and longevity; the lipidated GLP-1 molecules are bound to albumin, which is too large to be renally filtered, and are thus shielded from plasma proteases.
The combined effect of these modifications increase the drug’s half-life from minutes to approximately 168 hours, enabling its once-weekly injection schedule — an enormous improvement compared to the twice daily injection required for exenatide (Byetta) which was the first GLP-1 drug that was launched.1 Semaglutide (under the brand names Ozempic and Wegovy) swept the market. However, these modifications to the molecule could not solve the problem of degradation and poor absorption when taken by mouth.
From Silkworm to Trojan Horse
We might draw parallels for oral delivery of peptide and protein drugs from the case of serratiopeptidase, a little‑known drug from the 1960s with a peculiar history.
Serratiopeptidase is used to treat post‑operative inflammation in a handful of countries, including Japan, India and Sierra Leone. Produced naturally by the bacterium Serratia marcescens, which resides in the gut of the Japanese silkworm, this enzyme is used by the moth to dissolve silk fibers as it emerges from a cocoon. It is pharmacokinetically puzzling as an oral therapy because, as a peptide, it faces the usual hurdles of gastric degradation and poor epithelial uptake. Yet, surprisingly, no injectable formulation of serratiopeptidase is marketed. The reason is that intravenous administration carries a risk of immunogenicity — the immune system recognizes Serratia‑derived molecular signatures on the enzyme as foreign, which can trigger immune reactions. This danger is blunted or even absent, however, when the drug is given orally.
Serratiopeptidase only works because the oral dose is high enough that the small fraction of the drug which survives the gut reaches systemic circulation at concentrations sufficient for therapeutic effect. In this way, it serves as a proof‑of‑concept that even a peptide or protein drug known to have poor bioavailability can still be delivered orally, and without any modification, if we compensate by delivering a sufficiently high dose.
But in general, turning a peptide (including GLP-1 or insulin) into an oral formulation means overcoming three major challenges: protecting the peptide from digestion in the stomach, improving absorption of the peptide via the gut, and slowing down the enzymatic breakdown and excretion of the peptide once it is in the blood. Novo Nordisk’s oral semaglutide, Rybelsus, employs both co-formulation and clever biochemical engineering of the drug molecule itself to move us closer to this goal.
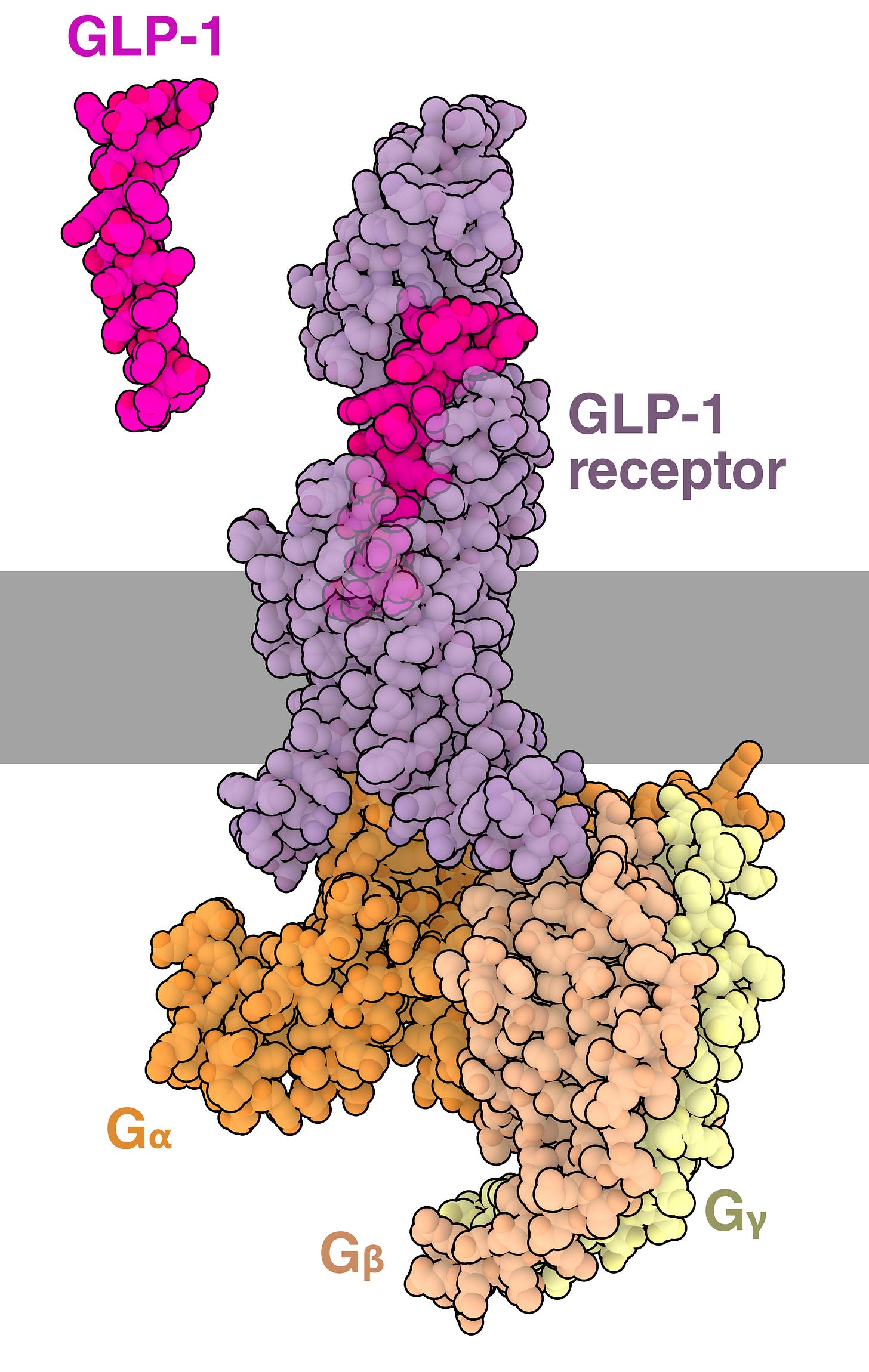
Specifically, Rybelsus works by co-formulating semaglutide with SNAC (sodium N-[8-(2-hydroxybenzoyl) amino] caprylate), a small synthetic molecule that functions as an absorption enhancer in the gastrointestinal tract. SNAC raises the pH of the immediate environment around the drug. The higher pH protects the peptide from pepsin degradation, which requires acidic conditions. SNAC also acts as a transient permeation enhancer, aiding semaglutide’s absorption into the blood, directly through the stomach lining and the small intestine.
While this co-formulation certainly improves oral delivery, it hasn’t fully solved the problem of dosage requirements. One pharmacokinetic study showed a modest bioavailability of only 0.8 percent for oral semaglutide, meaning less than one percent of the drug is absorbed into the blood where it can take effect.
To be effective, then, Rybelsus must be formulated to contain high doses of semaglutide. Whereas the GLP-1 drug Ozempic is injected once weekly at 2 mg, Rybelsus is taken daily and requires a minimum dose of 14 mg to achieve comparable glycemic effects. That is almost a 50-fold difference in total weekly intake for what is, fundamentally, the same drug, simply because most of the substance is degraded before they can be absorbed into the blood.
Additionally, Rybelsus doesn’t perform as well as its injectable counterpart, even at this higher dosage. In clinical trials, Ozempic (2 mg weekly subcutaneous) delivered a 15-20 percent body weight reduction over 68 weeks. Rybelsus at the approved 14 mg daily dose, though, achieved just 4-5 percent in the PIONEER trials. However, at 50 mg, oral semaglutide (OASIS 1) narrowed that gap substantially, achieving 15.1 percent weight loss at 68 weeks, approaching injectable performance.
While it is no secret that the cost of production of the drug molecule itself is only a small part of the retail price, the higher dosage required by oral delivery becomes a non-trivial difference in manufacturing costs.
Semaglutide costs $70,000 to $100,000 per kilogram to manufacture at commercial scale, according to two studies. Producing the drug ingredient contributes only $0.20 to the cost of each weekly 2mg injection. What forms the bulk of the retail price2 of peptides is not the cost of manufacturing, but the capital infrastructure: the regulatory burden, the cold chain logistics, the pen device (which sometimes costs more than the drug itself), and, above all, the return needed to justify $9 billion in the initial R&D of these GLP-1 drugs.
The cost breakdown looks much different for an oral semaglutide. In the OASIS 1 trial, patients took a 50 mg daily dose — meaning a monthly supply contains roughly 1,500 mg of active drug compound. A monthly injectable supply, by comparison, contains only about 8 mg. At $100,000 per kilogram, the drug compound alone for an oral monthly supply could already cost around $150, versus less than $1 for the injectable — before factoring in formulation, packaging, distribution, or profit.
This is the central problem with oral GLP-1s, then; biochemical engineering can technically solve the half-life problem, and co-formulations can improve the absorption problem, but the higher doses required make for formidable raw ingredient costs.3
Turning Food into Drug Delivery Vehicles
A solution to this problem may come from foregoing chemistry and turning, instead, to biology.
Genentech pioneered recombinant manufacturing of human insulin by cloning the human gene into E. coli as far back as 1978. Even today, many GLP-1 peptides are made by cloning the corresponding gene into microbes, culturing them in a fermenter, and then subjecting the resulting broth to successive rounds of filtration and chromatography to isolate the peptide precursor before undergoing chemical modifications. E. coli, though, is not edible, as it is laden with endotoxins that can trigger a septic shock. Therefore, any drug made in a cell like E. coli must be purified extensively, even if delivered orally. This downstream purification accounts for up to 80 percent of the total manufacturing cost.
Suppose, instead, that the microbes used for production were also edible. The drug could then be swallowed inside intact cells, and the massive cost of purification would disappear. In theory, there are millions of microbe species that can be engineered to synthesize peptides. But currently, we have the tools to genetically engineer only a few, and most microbes have never been studied for food safety and toxicology.
One attractive candidate is the edible, single-celled cyanobacterium Arthrospira platensis (spirulina). Cyanobacteria and many microalgae species have been consumed by humans for centuries and are Generally Recognized As Safe (GRAS) by regulatory agencies. Many people already eat spirulina for its antioxidants. Thus, the organism could be quite appealing for direct oral delivery in its intact cellular form, if it could be engineered to synthesize a GLP-1 molecule in sufficient quantities. Given the low cost of production, a 20-gram scoop of dried “algae powder” taken twice daily could, in principle, contain an orally adjusted therapeutic dose of a GLP-1 agonist for around $4/day.4

The cost argument alone is compelling, but using edible algae provides another benefit: keeping the GLP-1 inside the algal cell wall may improve the drug’s survival in the gut. Experimental data have already demonstrated that over 70 percent of the expressed proteins in spirulina remained intact after two hours of incubation under simulated gastric conditions that would fully degrade the same protein in a purified form within minutes. The cell wall acts as a natural enteric capsule, shielding the GLP-1 molecules from pepsin and the acid bath of the stomach, and releasing it progressively as it transitions into the higher-pH environment of the duodenum.
The reason this concept has not yet been tested in the context of GLP-1 is because, until very recently, it was not possible to engineer microalgae and cyanobacteria. Spirulina was, until a few years ago, considered genetically intractable because it has multiple copies of its genome and is resistant to standard transformation methods. In 2022, though, a paper reported successful engineering and high-level expression of therapeutic proteins in spirulina, with the encoded proteins accumulating to up to 15 percent of total dry biomass.
Today, newer organisms like UTEX 3222 (discovered in 2023 and also known as “Chonkus”) can double several times faster than spirulina and be engineered to express recombinant peptides. Some of these strains are quite simple to grow, needing just light and CO2. Their adoption might not only make oral GLP-1s affordable, but also allow the mass production of customized peptides from photosynthesis — all inside edible cells.
Many cyanobacteria (and microalgae broadly) are safe to eat, though not every strain of cyanobacteria has a GRAS certification.5 Under the GRAS framework, an ingredient has to be demonstrated as safe either “through (1) scientific procedures or (2) in the case of a substance used in food prior to January 1, 1958, through experience based on common use in food.” For cyanobacteria to be used as a platform for oral GLP-1 delivery, either a GRAS organism like spirulina must serve as the host organism or, if choosing a different species, the manufacturer must prove with adequate in vivo trial data that it is safe to eat.
In addition, one must gain FDA approval by clinically demonstrating that the delivery of the microbe-encapsulated GLP-1 through this oral route leaves the drug molecule itself unchanged (with no post-translational modifications), that it has a similar pharmacokinetic profile to the reference molecule, and that it shows no adverse effects. Since this is a new delivery platform of existing drugs rather than a new drug, the regulatory bodies in many jurisdictions typically have alternative, shorter approval pathways available to drug sponsors that do not require the standard Phase I-III clinical trials for a new drug application.
A third argument for adopting edible organisms for GLP-1 delivery would be to increase global accessibility. Bangladesh, for example, ranks among the world’s top ten countries for T2DM patient burden, with a prevalence of 9.2 percent among adults. The disease currently impacts over eight million people, a number projected to grow to 15 million by 2045. Yet GLP-1 drugs are effectively absent from the formularies of Low and Middle Income Countries (LMICs). A 2024 study found that GLP-1 drugs can likely be manufactured more cheaply than their current market costs of, for example, $95 in Brazil, $115 in South Africa, and $353 in the United States.
Médecins Sans Frontières has called for Novo Nordisk and Eli Lilly to license their patents to generic manufacturers, noting that neither company has adopted pricing strategies for low-income markets. The surge in U.S. demand has also triggered global shortages that hinder the drug’s availability for essential diabetes care in LMICs. If we can provide alternative biomanufacturing capacity that slashes cost, turns injectables into safe oral, algae-encapsulated formulations, and eliminates the need for refrigeration, we may be able to ensure that patients in LMICs are no longer last in line for the latest and best therapeutics.
So far, three major problems have kept GLP-1 drugs injectable rather than oral: enzymatic degradation, poor bioavailability, and the sheer cost of manufacturing. Novo Nordisk's Rybelsus showed that transcellular gastric absorption, boosted with SNAC, can partially get around the first two problems, but cannot solve the third. This is why research groups around the world are trying to engineer microbes to act as edible drug factories. The idea is to skip the most expensive part of manufacturing, downstream purification, by keeping the expressed peptide locked inside the protective walls of edible cells.
The science of oral peptides, at this point, is no longer a mystery. We know oral peptide drugs can work. The harder question is whether patients will come to accept the idea of eating their medicine in a spoonful of algae.
David S. Kim is the founder of CyanoCapture [General Bio], a synthetic biology company that is manufacturing peptides by genetically engineering non-model microbes. He is a medical doctor based in the U.K.
Cite: Kim, D.S. “The Quest for Oral GLP-1s.” Asimov Press (2026). DOI: 10.62211/82ej-91yt
Liraglutide has a C16 palmitoyl fatty acid at lysine-26 via a short linker, binding albumin, extending its half-life from under 2 minutes to 11-15 hours, now allowing for once-daily dosing. Semaglutide has a C18 di-acid fatty chain attached at the same lysine-26 position, connected through a hydrophilic linker comprising two mini-PEG units (OEG) and a gamma-glutamic acid spacer, confers albumin affinity 5.6-fold greater than liraglutide’s.
Brand-name Rybelsus lists at around $997/month in the U.S. Ozempic and Wegovy are priced between $500-1,800/month without insurance. Popular U.S. telemedicine platforms undercut this dramatically by using compounded semaglutide: Mochi Health charges $99/month for compounded injectable semaglutide; Hims offers compounded semaglutide from $199/month; Ro’s GLP-1 programs start at $349/month.
The emergence of small-molecule GLP-1 receptor agonists represents a possible inflection point. Orforglipron, Eli Lilly’s oral non-peptide GLP-1 agonist currently awaiting approval, is synthesized by conventional organic chemistry in a standard chemical reactor. Manufacturing costs are estimated at 30-50 percent below those of peptide-based competitors.
This figure assumes $100/kg of producing dry cyanobacteria biomass containing a therapeutic protein under cGMP conditions and 2 x 20g of powder required to deliver the oral adjusted dose of GLP-1 peptide.
UTEX 3222 is not GRAS yet as it was only discovered in 2023. It is in the pipeline for receiving GRAS certification. Spirulina (cyanobacteria) is GRAS, and that is what some companies (Lumen Biosciences) are using to make oral biologics. Chlorella and Chlamydomonas are also well known examples of GRAS species of microalgae (though not cyanobacteria) commonly found on supermarket shelves.

I thought the use of cyanobacteria is a really interesting idea. As a plant synthetic biologist, I'm all for using biology and the power of photosynthesis to make high value products (aka, molecular farming). However, in this case, I'm curious: Why use spirulina or other non-model organisms over something like yeast which we eat everyday, is highly tractable genetically, and is already established in biomanufacturing?
I'm also curious how much research is still being done to make these peptides more stable in the gut (probably a lot). I've heard cyclizing peptides can be a way to stabilize them, so I wonder if that's being tried.
What fascinates me here is the engineering mindset behind GLP-1 drugs. The biology was known decades ago, but the real breakthrough came from chemical tweaks that stretched a molecule’s lifetime from minutes to days. Sometimes innovation is less about discovery and more about persistence in optimisation.