Metaphors for Biology: Evolution
A series of quantitative metaphors on the speeds of representative events in evolutionary biology. The end of our three-part series.
To determine which is faster, natural or artificial selection, one might select examples to compare: while wolves and wild jackals branched off from a common ancestor 3.5 million years ago (natural selection), arctic foxes were domesticated within a couple of decades (artificial selection). The oldest moth species appeared in the fossil record around 200 million years ago (natural selection), while the peppered moth changed from a light-colored to a primarily dark-colored one in just 47 years (artificial selection).
But this becomes harder when we consider questions like: Is COVID well-adapted to human hosts? What animal or plant has had as much time to adapt to a new environment as COVID has to humans? Is the persistence of dominant genetic disorders best explained by a lack of purification time (i.e. that humans haven’t lived in their current environment long enough to purify out harmful alleles that might have once been adaptive) or something else?
Very quickly, two major issues arise. First, “years” seems an inaccurate unit for expressing evolutionary rates because evolution considers changes between one generation and the next. In absolute terms, dog, moth, and COVID-19 generations occur over different spans of time; yet from the perspective of evolution, all three should probably be treated similarly.
A second important consideration is that “the time it takes to evolve [X]” is a parameter with large, inherent error bars. It’s difficult to measure rates of evolutionary change accurately, and biological evolution is an inherently stochastic process, contingent on events like a cell accidentally skipping a base in DNA replication or a tortoise, swept away in a storm, happening to land on an island full of edible ferns. Without such random events, an evolutionary process that took a million years might have taken half or twice as long.1
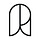
Still, we can get a feel for how long different kinds of evolutionary events take. Let’s standardize generation time and imagine one generation happens per second. Choose an organism and start a stopwatch. Each second, replace the individual in your head with its offspring. Imagine that offspring a tad different from its parents. Do this for thirty seconds, then a minute. How does that span feel? Can you picture evolution happening?
Let’s calibrate this clock against human history:
At one generation2 per second, the industrial revolution began ten seconds ago. There were fewer than a billion humans, the vast majority of whom were farmers.
If we go back seven minutes further, humans have just begun implementing large-scale agriculture.
Go back half an hour and behaviorally modern humans appeared. They possessed paints, composite tools, art, burial, complex hunting and fishing methods, and other common features of modern “primitive” civilizations.
Now let’s look at evolution itself, starting with some familiar examples and expanding to cover much of the eukaryotic tree of life. Then we’ll contrast these with examples of artificial selection to get a feel for their relative speeds.
Natural Selection
Human Evolution
Anatomically modern humans appeared about three hours ago. These are hominids who are essentially physiologically indistinguishable from contemporary humans.
Ancestors of Homo sapiens diverged from the other great apes (specifically, from the ancestors of modern gorillas and chimpanzees) some three and a half days ago. These were still primarily quadripedal apes.
The dinosaurs died out and mammalian diversity began to explode about one hundred days ago.3
Human-Pathogenic Viruses
Using the time from initial infection of a host to the successful propagation to the next host as “one generation”:4
Flu
Human influenza viruses experience approximately 19 minutes of evolution each season.
The influenza virus responsible for the devastating 1918 epidemic that killed some 1-5% of the world’s population still exists, having evolved over some 33 hours to become today’s H1N1 seasonal flu virus.
SARS-CoV-2
Under some simplifying assumptions5 we can estimate the time it took to evolve the major branches of SARS-CoV-2:
The Alpha strain was first detected 42 seconds after the first reported COVID-19 case. It peaked as a proportion of global cases at 72 seconds, then was gradually replaced and effectively went extinct after about 110 seconds.
Delta appeared about 66 seconds into the pandemic. It was a parallel lineage, not related to Alpha, which became the dominant strain worldwide after 2 minutes and rendered Alpha functionally extinct by the 2.25 minute mark.
The ancestor of all Omicron lineages probably first appeared 67 seconds into the pandemic. It was not derived from either Alpha or Delta, and wasn’t particularly successful itself, but it did give rise to both Omicron variant 21K and Omicron variant 21L (both first detected at 3 minutes). 21K displaced Delta and peaked at about 3.15 minutes and was displaced in turn by 21L another 10 seconds later.
In total, SARS-CoV-2 has existed in humans for approximately 10 minutes.
Evolution of Major Clades
The time to go from single-celled organisms to multi-cellular organisms is tough to estimate — single-celled organisms have generation times as fast as tens of minutes or as slow as decades. Assuming the most historically-important single-celled organisms were like today’s marine phytoplankton,6 multicellularity took 26,000 years to first evolve.
To understand the evolutionary age of the vertebrates, we might use generation times taken from one of the oldest extant vertebrate lineages — sharks. One study of shark reproductive parameters found average generation times for 18 shark species ranging from 3 years to 42 years, which implies a metaphoric evolutionary age of 140 days to 5.5 years.
Fast-maturing plants, reproducing annually, have been around for 15 years of evolution. Flowering grasses and other annuals have been around for 4.4 years, while typical flowering trees have evolved for only 1.5 years.7
What about fungi? The fastest-growing eukaryotes known to science (as of 2009) is an ascomycetous yeast called Kluyveromyces marxianus, which grows a bit faster than one generation per non-metaphorical hour. This sets an upper bound of the metaphorical age of all fungi at about 375,000 years (fungi are quite ancient). Other single-celled fungi like yeasts and slime molds have probably evolved for a few years; more familiar mushroom-producing fungi may have “only” been around for 40-odd years.
Artificial Selection
So far we’ve only covered examples of natural selection. But anything that selectively influences which organisms do and don’t reproduce can force an evolutionary change. Humans do this all the time by selectively breeding animals and plants for traits we like.
How does the speed of artificial selection compare to that of natural selection? We can start with a classic textbook example of evolution that bridges the two: the evolution of the peppered moth under unintentional selection pressures caused by the industrial revolution.
Before the industrial revolution, peppered moths living in Manchester, England were light-colored moths with a dark, splotchy pattern perfect for blending into forests. During the industrial revolution, coal soot and sulfur dioxide emissions from the city blackened the trunks of those trees, making peppered moths stand out. Under intense bird predation, the peppered moth switched from dark splotches on a light background to a totally dark coloration that allowed them to hide on soot-blackened trees. This rapid evolution took about 47 seconds.
Agriculture
The longest-running experiment in artificial selection is the domestication of crop plants.8 Crops are a particularly rich example of evolution because domesticated plants tend to pick up the same cluster of traits — non-shattering seed hulls, enlarged seed or fruit, shorter height, synchronized flowering — so the collection of all agricultural crops can be viewed as replicates in a single giant experiment in artificial selection.

To get a sense of how crop domestication works, let’s look at the recent evolutionary history of a crop for which we have particularly complete evidence: barley. Barley was among the most utilized crops in early agricultural old-world societies, grown across nearly all of Eurasia and North Africa. Critically, it is one for which we have an abundance of archaeological evidence. It seems to have been an unusually slow crop to cultivate, so the evolutionary times given here for barley are more representative of the relative pace of domestication than of its absolute time.
A brief and vastly simplified9 history of the domestication of barley,10 in our metaphorical time, would be that:
The oldest evidence of cultivation of phenotypically wild barley appeared approximately 20,000 years ago, and serves as our zero second mark.
The first recognizably cultivated barley appears at around 2.7 hours, after almost 10,000 generations. These seeds look a lot like wild barley, just with bigger grains.
The first recognizably cultivated barley slowly spreads throughout the fertile crescent over the next 50 minutes.
Meanwhile, barley gains a “non-shattering hull” just 7 minutes after first displaying a larger grain. This variety has a more solid seed husk, less prone to spontaneously breaking open and prematurely scattering its seeds before harvest.
The first 6-row barley appears just 10 minutes after the first non-shattering hulls. Wild barley bears two seeds at each repeating subunit; this variety bears six instead, greatly increasing yield.11 (We are now about 3 hours into domestication).
A second, independent domestication event occurs in Asia around 3 hours and 12 minutes in.
The first hulless barley appears in Iran just a few minutes later, at the 3 hour and 15 minute mark. “Hulless” is something of a misnomer as this barley still has a hull. However, it is much, much easier to remove. Both hulless and hulled barley will continue to be used for different purposes up through the present day.
A “dense 6-row” barley appears at about 4 hours and seven minutes, bearing more seeds per ear.
At about 4 hours and 20 minutes, Hulless barley begins to appear much more broadly across Europe.
Barley cultivation continues with shifting balances and mixes of existing varieties until close to the present day, around five and a half hours in.
In the last minute of barley cultivation, humans have modified barley to grow a shorter stalk (less prone to breaking under heavy seed load) with greater seed-to-vegetation weight ratio, and increased resistance to several important diseases.
Barley domestication teaches us that the process occurs in many steps, spread out over thousands of generations; both wild and cultivated varieties were used virtually unmodified for thousands of generations before undergoing multiple major changes over just a few hundred. New traits take almost as long to spread and take over cultivated populations as they take to arise in the first place. Individual domestication events seem to require on the order of thousands of generations, not hundreds and rarely tens of thousands.
But the real value of agriculture as a case study in evolution is the number of replicates it provides. Ideally, to get the best feel for the timescale of plant evolution under artificial selection, we would re-visualize domestication for each of the thousands of species humans have domesticated as crops.
Luckily for us, Meyer, DuVal, and Jensen have collected evidence of domestication for 203 food crops from around the world into a handy spreadsheet. Limiting ourselves to crops with high-quality evidence, we can see that:
The median crop becomes domesticated after about 24 minutes of (metaphorical-time) exploitation by humans, while mean domestication time is a bit longer, at 33 minutes.
The fastest-domesticated crop is the common bean, for which the archaeological evidence for first exploitation and first domestication are indistinguishable (though this may say more about the quality of our evidence than the friendliness of the common bean).
The most recalcitrant crop to eventually yield to domestication is, ironically enough, barley, at its rather lengthy 2.7 hours between first exploitation and eventual domestication.
Annuals — crops with a one-year lifecycle — take somewhat longer to domesticate, with a median domestication time of 33 minutes.
Perennials — crops that live more than one year — domesticate more quickly, with a median domestication time of just over 9 minutes.
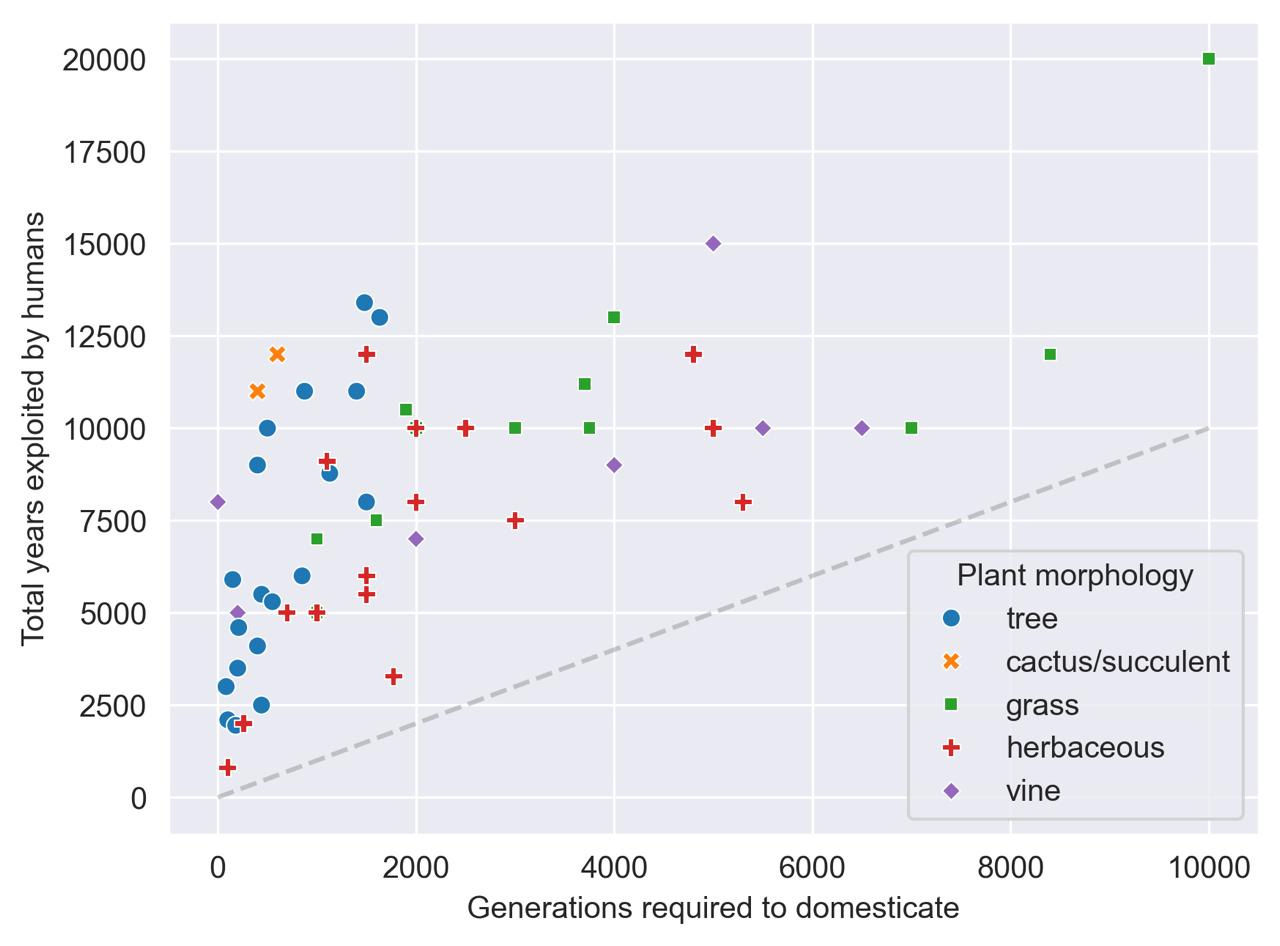
We can do a lot with this data. For example, if we split up domesticated crops by their gross morphology,12 it appears that trees (and possibly cactuses and succulents) may take fewer generations to domesticate than grasses, vines, and other herbaceous crops.13
Specifically, on average:
Trees take about 10.5 minutes to domesticate.
Cactuses and succulents take 8 minutes to domesticate (but N=2).
Grasses take a bit over an hour to domesticate.
Vines take about 40 minutes to domesticate, while herbaceous plants take about 35 minutes to domesticate.
Domesticated Mammals
The evolution of the dog from wolf-like ancestors bridges a gap between crop domestication and pure artificial selection. It began as an unintentional and gradual process, but advances in selective breeding (and in the scientific understanding of evolution itself) led to a recent acceleration in dog “speciation.” Its history is also complex, speculative, and contentious, but we have enough combined genetic and archaeological evidence to broadly estimate that:
The ancestors of dogs split from modern wolves anywhere from 2.5 hours ago to 50 minutes ago by wolf generations,14 or 2-5.5 hours ago by dog generations.15
The earliest recognizably domesticated dog remains date to approximately 2 hours ago.
Most modern dog breeds — especially hunting breeds like greyhounds, schnauzers, terriers, beagles, and bloodhounds — are thought to have split from a common ancestor only 1-2 minutes ago.
We also have much more intentional, directed examples of mammal domestication. In 1952, Soviet geneticist Dmitry Belyayev and his graduate student Lyudmila Trut began a multi-decade experiment to attempt to replicate the domestication of dogs. Instead of wolves, Belyayev chose silver foxes as the base species for domestication, starting with foxes bought from commercial fur farms.
Generation after generation, Belyayev and Trut separated out the young foxes who responded most positively to human handlers, as well as the foxes who responded most aggressively. By breeding the most sociable foxes together, the scientists obtained increasingly friendly, loyal, and loving individuals; conversely, the antisocial-selected line quickly evolved heightened aggression responses.
The domestic fox experiment continues to this day. Some key observations are that while the researchers initially categorized their foxes into three “classes” based on their level of tolerance to approach by humans, foxes soon appeared that actively sought human attention, requiring a fourth “elite” category.
The first four of these “elite” foxes were born after 6 seconds.
After 30 seconds, fully half of the foxes were born elite.
After 42 seconds, almost three quarters of friendly foxes were elite.
As early as 20 seconds into the experiment, some of the domesticated foxes started to be born with dog-like physiological traits, including floppy ears, piebald coat patterns, shortened or upward-curled tails, and shorter legs.16
Domestication was also associated with a dramatic decrease in corticosteroid production, which the experimenters hypothesize is responsible for the foxes lack of fear response towards humans. By 30 seconds, the domestic foxes had half the baseline cortisol blood concentration of un-selected controls (a ratio which hasn’t changed much since).
The same experiment has been repeated in other species:
River otters showed early signs of domestication after one or two generations, but breeding them proved too difficult to sustain and their breeding was cut short.
Wild Norwegian rats (“laboratory rats”) adapted so quickly over their first 35 seconds of breeding that they maxed out the researchers’ initial behavioral scale and forced them to define a second one.17
American minks began showing active interest in human handling after 18 seconds of selection.
Evolution in the Lab
The most direct and well-quantified examples of human-induced evolution come from laboratory experiments on single-celled bacteria and protists. Fast-growing, single-celled organisms make amazing test platforms for evolution, due to their ease of genetic manipulation and characterization and incredibly fast reproduction. One test tube, incubated overnight, can easily hold tens of billions of E. coli, representing millions of de novo genomic mutations. On the other hand, bacteria reproduce asexually, without sexual recombination, which makes even huge populations of bacteria less genetically diverse than, say, a small tribe of monkeys.18
With those features in mind, let’s look at how scientists have measured — and forced — evolution in microorganisms. In a famous long-term evolution experiment in the lab of Richard Lenski, twelve parallel lineages of laboratory-bred E. coli have been successively grown in simple, glucose-limiting media for over 35 real-world years and 80,000 generations — a bit over 22 hours of metaphorical time. A few highlights from that experiment, in metaphorical timescales are that:
The E. coli evolved very quickly at first, then slowed considerably (but never stopped). In the first half-hour of evolution, each of the 12 strains increased their fitness in their new environment by about 40 percent.
One of the first real surprises of the experiment was the sudden appearance of extreme mutation rates in 6 out of 12 strains. These “hypermutator” strains are estimated to be about 1 percent less fit than their more stable ancestors when they first appear (because they are much more likely to replicate genomes with harmful mutations), but were able to evolve more quickly and therefore were able to outcompete their ancestors in the long term.
The hypermutators took over their respective populations at about 40 minutes, 45 minutes, 1.4 hours, 2.4 hours, 7.5 hours, and 9.7 hours into the experiment.
One — and only one — of the twelve E. coli strains evolved the ability to eat citrate from their growth media after about 31,000 generations, or 8.6 (metaphorical) hours of adaptation to lab conditions. This trait was dependent on a set of background mutations on acetate metabolism, which arose somewhere around 7 (metaphorical) hours into the experiment.19
These first citrate-eaters were pretty messy eaters that spewed carbon-rich metabolites20 as a by-product of citrate metabolism. Within about 15 minutes (900 generations) of their appearance, another non-citrate-metabolizing lineage evolved a greatly-enhanced ability to consume those by-products.
The citrate-metabolizing and by-product-metabolizing strains co-existed for about 2.7 hours of metaphorical time, until the citrate-eating E. coli got good enough at consuming their own waste products to out-compete the secondary strains and drive them to extinction.
The E. coli evolved to grow steadily larger over time, roughly doubling in cell volume in the first metaphorical half hour of the experiment. They continued to grow in size over the rest of the experiment, but more slowly, expanding to 2.5x their ancestors’ original volume after about 2.7 hours and 3.5x the ancestral volume after 14 hours.
Other examples of long-term evolution experiments abound, too. In 1998, for example, Boraas, Seale, and Boxhorn forced (or at least strongly encouraged) the single-celled green algae Vulgaris chlorella to evolve to grow in multi-celled clumps by exposing them to heavy predation by a flagellate predator. Under this intense selection for larger, harder-to-eat forms, V. chlorella evolved into eight-celled clumps in 10-20 (metaphorical) seconds.
And in a more recent example, four scientists at the University of Minnesota used an even simpler, more ingenious method to select for multicellularity — they propagated media containing Saccharomyces cerevisiae (baker’s yeast) from the bottoms of test tubes over many generations.21 This artificially selected for yeast growing in many-celled “snowflakes” that could sink quickly.22 In our metaphorical time scale, out of ten parallel replicates of evolved yeast, one population evolved noticeable numbers of such snowflake yeast in about 1.5 minutes, half evolved them by 2.5 minutes, and all ten were “multicellular” in 6-7 minutes. The same experiment has been replicated a number of times on a number of different single-celled eukaryotes, with different rates of evolution:
70 seconds for Kluyveromyces lactis (another budding yeast that diverged from S. cerevisiae about 100 million non-metaphorical years ago).
2.5 minutes for Schizosaccharomyces pombe (a fission yeast).
Between 1.5 and 7 minutes for Sphaeroforma arctica (a single-celled protist relatively closely-related to animals).
In slightly over 5 minutes, one out of twelve replicates of Chlamydomonas rheinhardtii (a single-celled green algae closely-related to plants) evolved multicellular clumping.
Trichormus variabilis (a cyanobacteria) increased in size (through a combination of larger cell size and clumping) by about 3-fold over 2.5 minutes and >30-fold over 6 minutes.
Feeling Evolutionary Time
We’ve argued in previous articles that metaphor helps with quantitative understanding of the sizes and speeds of molecular biology. But metaphor really becomes useful when considering evolutionary time. Relating the number of generations required for evolutionary events to everyday time scales helps us see patterns in what would otherwise seem like totally incomparable evolutionary facts.
Our examples have shown us that intense artificial selection can drive changes far faster than natural selection. “Natural” domestication of both plants and mammals seems to act on the metaphorical scale of hours (with some exceptions, like trees and cacti!), but intense selective breeding can create dozens of new dog breeds in minutes, or even recapitulate dog-like domestication in foxes in tens of seconds. The intensity of selection is just as, or more, important than the size of the difference produced, when it comes to how long it takes to evolve a change.
We have also seen that bacteria evolve relatively slowly. Half a dozen different single-celled eukaryotic species were capable of evolving primitive multicellularity in a matter of metaphorical minutes; E. coli, in contrast, adapted to experimental conditions more on the order of tens of minutes or hours—even though they could draw on absolutely massive population sizes! Speculatively, this may be because eukaryotes use sex (and other powerful DNA-recombination tricks) to generate lots of genetic diversity even from relatively small populations.
And we’ve learned that evolutionary speeds are trends, not exact rates. Several examples (plant domestication, hypermutators in E. coli, multicellularity in single-celled eukaryotes) reveal that roughly the same change occurs multiple times in different populations or species. When this happens, the speeds of the individual examples vary, but only about 3- to 5-fold around the median speed.
We’ve seen that the speed of evolution is complicated. It depends on the size of the evolving population, the strength and stringency of selection, and how the species reproduces—and how quickly. The “one generation per second” quantitative metaphor can’t eliminate all of that complexity, but it at least puts species with radically different life cycles on an even footing, which helps clarify where and when other factors like population size or replication fidelity come into play.
More importantly, using a single scale of “one second per generation” lets us place concrete evolutionary examples onto a more intuitive reference frame. The next time you learn that a weird animal lived so many millions of years ago, or a bacteria evolved antibiotic resistance in so many days, you can place it on that same common scale — simply divide the total time by the length of one generation, convert that number of seconds into something convenient like minutes or days, and you’ll be able to compare it directly to any of the examples given in this brief introduction.
Hopefully, this technique has given you a better sense of how evolution works.
Samuel Clamons is a bioinformatics scientist at Illumina, Inc. with a PhD in Bioengineering and training in applied mathematics and computer science. Outside of his day job, he writes science fiction and researches theoretical questions in biology at Asimov Press.
Many thanks to Dr. Scott Biering and Dr. Alistair Russell of UCSD for their consultations on viral reproduction and evolution. Header image by Ella Watkins-Dulaney. Image Credits: M. Garde and Tkgd2007.
Cite: Clamons, S. “Metaphors for Biology: Evolution.” Asimov Press (2026). DOI: 10.62211/29je-83ut
Even in explaining this subtlety, I’m ignoring an important and fascinating tension at the heart of the mathematics of evolution. Evolution requires two steps best described with fundamentally different kinds of math: first, a trait must appear in a population; then, the trait must change in frequency in the population (e.g., spread and take over).
The appearance of traits typically depends on a mutation, which is a totally stochastic process. Every time an individual reproduces, it has some chance of gaining the mutation. This is the most noisy, random part of the process, with inherent “error bars” that are just as important to understand as their average rates.
Once a trait exists, its fate is still subject to chance. An individual with a new trait, even a highly adaptive one, might happen to be eaten by a predator as an egg, larva, or adolescent. It might be caught in a rockslide, catch a deadly illness, or form a life-long pair bond with a sterile mate. It might be born with a debilitating or lethal trait unrelated to the new, advantageous one. Or it might stumble across a rich, safe new ecosystem where it can breed like crazy. The fate of the new allele isn’t, for the most part, in the hands of that allele!
But if the new trait gets a chance to spread, then all those dice rolls start to average out. While one individual with a new allele could easily die randomly, five siblings carrying the same trait most likely would not. If the population is sufficiently large and the new trait takes over a sufficient fraction of it, evolution stops acting like dice rolls and starts acting more like billiard balls with predictable trajectories — its dynamics become deterministic. The spread of the new trait through the species begins to occur according to predictable timescales.
Each evolutionary example we’ll cover is a blend of stochastic and deterministic processes. Some examples’ rates are determined mostly by the stochastic part of evolution, while others are dominated by the deterministic. Thinking through the weighting for each example will make your mental evolutionary judgment more accurate.
Using a figure of 27 years per human generation, estimated from historical mutation rates.
This is a tricky estimate because mammals that far back might have had quite different generation times. I’ve assumed an average generation time of 9.5 years, based on the average generation time across 418 primates (primates probably split from other mammals just before the K-Pg extinction, geologically speaking), and I’ve rounded the answer to one significant figure.
We use inter-infection time as our measure of “a generation”—instead of, say, the time it takes a virus to replicate its genome, or the time it takes an infected cell to begin releasing new viral particles—because infection represents a strict, extreme reproductive bottleneck in a virus’s life cycle. A virus might replicate its genetic material billions or trillions of times during the course of an infection, but at most only a few dozen of those genomes will actually go on to successfully infect another host—much the same way a human replicates their genetic material hundreds of trillions of times, yet only passes on their genes in a living descendent a handful of times.
Namely: that the SARS-CoV-2 virus spends very little time in the environment between infections (usually reasonable, although there have been documented cases of SARS-CoV-2 being transmitted after sitting on cold food packages for days or months); and that all SARS-CoV-2 transmission is human-to-human (definitely not true—the virus is known to jump from animals to a number of species and back again—but unfortunately necessary for this analysis, as we know far less about the virus’s replication cycle in non-human animals).
Which reproduce about once each non-metaphorical day.
They’ve been around subjectively longer than dinosaurs because of the 1-year generation time assumption; in absolute, non-metaphorical time, flowering plants only showed up in the Cretaceous period, the third and final period of the dinosaurs.
This section draws heavily from “Patterns and processes in crop domestication: an historical review and quantitative analysis of 203 global food crops” (2012), by Rachel S. Meyer, Ashley E. DuVal, and Helen R. Jensen, New Phytologist 6(1). The paper’s supplemental data, containing a summary table of 203 crop species and their uses, age and extent of domestication (Table S5), is a particularly rich data source.
Barley, like most crops, isn’t one species with a linear history—it’s a many-branched and tangled family tree of closely-related cultivars, each with its own timeline of trait adoption. The numbers here are for the first appearance of each trait, in any cultivar.
Still using Meyer 2012 as a main source, along with data from “Contrasting Patterns in Crop Domestication and Domestication Rates: Recent Archaeobotanical Insights from the Old World” (2007), by Dorian Fuller, Annals of Botany 100(5).
Six-row barley virtually completely replaced 2-row barley until the 16th century, when 2-row came back into use in some places. Two-row barley has also made a recent comeback as a superior beer-brewing crop, due to various favorable differences in its biochemistry.
Not to be confused with evolutionary relatedness. Don’t forget, trees aren’t real.
According to a Mann-Whitney U-test, trees are statistically significantly faster to domesticate than non-tree crops (p = 0.00011). Make of that what you will.
Using an average generation time of 4.5 years, as measured by Mech & Erb (2016).
Dogs reach sexual maturity about twice as quickly as wild wolves, and can be bred earlier.
A small fraction, but still several orders of magnitude larger than seen in the original, basal population.
The initial scale measured aggressive responses to human handling. By 35 generations, no rat displayed any aggressiveness, and the experimenters switched to measuring the degree to which the rats felt comfortable exploring and playing in their handlers’ presence.
Yeasts and many single-celled protists can reproduce sexually, but most prefer to clonally bud or split, especially when conditions are good.
The citrate metabolism mutation involved a duplication of a previously-dormant citrate-transporting gene that exposed one of the two copies to a nearby active promoter, effectively switching it on. When Lenski-lab researchers intentionally added this mutation into recent ancestors of the citrate-eating E. coli, the engineered bacteria also gained the ability to use citrate; when they added the mutation into more distant relatives that had diverged from the citrate-eating lineage before the 7-hour mark, it had very little effect.
C4-dicarboxylates. Specifically, succinate, fumarate, and malate.
They first centrifuged their tubes to speed up the settling effect of gravity.
They also developed rudimentary division of labor and what looked like programmed cell death. Snowflake yeast cells near the center of a growing flake eventually evolved to intentionally commit suicide by apoptosis, allowing the snowflake as a whole to grow faster and possibly to “reproduce” (by shedding smaller snowflakes) more rapidly.

The banner image used of an ape linearly transitioning to a human is wrong and misleading. Instead the metaphor of a tree with branching from common ancestor should be used. This is so widespread thanks to Google bringing this up image as the first hit. But I am surprised that a good article on evolution should use this misleading figure. Such usage gives credibility to the wrong image and further propagates it. Do change if possible and also refrain from using this linear image